|
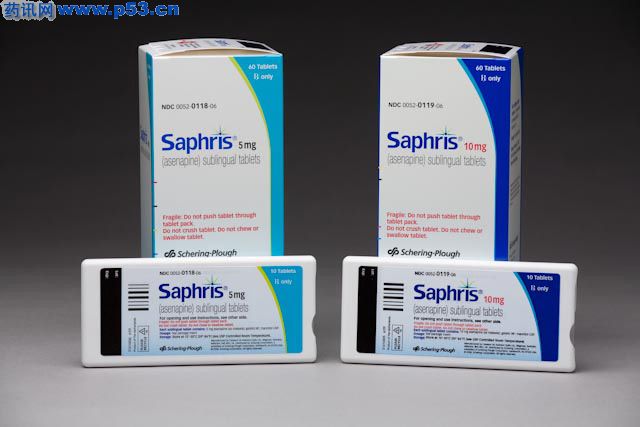
SCHERING-PLOUGH ANNOUNCES FDA APPROVAL OF SAPHRIS® (ASENAPINE) FOR ACUTE TREATMENT OF SCHIZOPHRENIA IN ADULTS AND MANIC OR MIXED EPISODES OF BIPOLAR I DISORDER WITH OR WITHOUT PSYCHOTIC FEATURES IN ADULTS "We are very pleased with the U.S. approval of SAPHRIS, which represents an important new choice for acute treatment of schizophrenia and acute manic or mixed episodes of bipolar I disorder in patients starting treatment and those who have discontinued previous treatment," said Thomas P. Koestler, Ph.D., executive vice president and president, Schering-Plough Research Institute. "SAPHRIS is an important addition to Schering-Plough's product portfolio, and represents the first U.S. approval resulting from the Organon/Schering-Plough combination." SAPHRIS is expected to be available in the U.S. during the fourth quarter of 2009. Schizophrenia affects about 24 million people worldwide, including two million Americans, and bipolar I disorder affects about 1 percent of adults, including 10 million Americans. "Schizophrenia and bipolar I disorder are complex medical conditions that can present clinical challenges for the physician," said Steven G. Potkin, M.D., professor, department of psychiatry and human behavior, University of California, Irvine, and lead author of a pivotal schizophrenia study as part of the SAPHRIS clinical development program. "Having a new FDA-approved treatment such as SAPHRIS is important in these serious conditions because physicians need options to help manage their patients' symptoms," said Roger S. McIntyre, M.D., associate professor of psychiatry and pharmacology, University of Toronto, Canada, and lead author of the pivotal bipolar mania studies as part of the SAPHRIS clinical development program. The FDA approval of SAPHRIS is based on a New Drug Application (NDA) that included efficacy data from a clinical study program involving more than 3,000 patients in schizophrenia and bipolar mania trials. The SAPHRIS filing was supported by safety data in 4,500 people, with some patients treated for more than two years. The approval is based on acute schizophrenia trials in which SAPHRIS (5 mg twice daily) demonstrated statistically significant efficacy versus placebo and acute bipolar I disorder studies in which SAPHRIS (10 mg twice daily) demonstrated statistically significant reduction of bipolar mania symptoms versus placebo. About SAPHRIS (asenapine)
In Europe, a Marketing Authorization Application (MAA) for asenapine, under the brand name SYCREST®, is currently under review by the European Medicines Agency (EMEA) for the treatment of schizophrenia and manic episodes associated with bipolar I disorder. The application will follow the Centralized Procedure. Schering-Plough acquired asenapine in November 2007 through its acquisition of Organon BioSciences, which developed the product. Important Safety Information about SAPHRIS Increased Mortality in Elderly Patients with Dementia-Related Psychosis: Cerebrovascular Adverse Events: There was a higher incidence of cerebrovascular adverse reactions (cerebrovascular accidents and transient ischemic attacks) including fatalities compared to placebo-treated subjects. SAPHRIS is not approved for the treatment of patients with dementia-related psychosis. Neuroleptic Malignant Syndrome (NMS): NMS, a potentially fatal symptom complex, has been reported with administration of antipsychotic drugs, including SAPHRIS. NMS can cause hyperpyrexia, muscle rigidity, altered mental status, irregular pulse or blood pressure, tachycardia, diaphoresis and cardiac dysrhythmia. Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis) and acute renal failure. Management should include immediate discontinuation of antipsychotic drugs and other drugs not essential to concurrent therapy, intensive symptomatic treatment and medical monitoring, and treatment of any concomitant serious medical problems. Tardive Dyskinesia (TD): The risk of developing TD and the potential for it to become irreversible may increase as the duration of treatment and the total cumulative dose increase. However, the syndrome can develop, although much less commonly, after relatively brief treatment periods at low doses. Prescribing should be consistent with the need to minimize TD. If signs and symptoms appear, discontinuation should be considered. Hyperglycemia and Diabetes Mellitus: Hyperglycemia, in some cases associated with ketoacidosis, hyperosmolar coma or death, has been reported in patients treated with atypical antipsychotics. Patients with risk factors for diabetes mellitus who are starting treatment with atypical antipsychotics should undergo fasting blood glucose testing at the beginning of and during treatment. Any patient treated with atypical antipsychotics should be monitored for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia and weakness. Patients who develop symptoms of hyperglycemia during treatment with atypical antipsychotics should also undergo fasting blood glucose testing. In some cases, hyperglycemia has resolved when the atypical antipsychotic was discontinued; however, some patients required continuation of anti-diabetic treatment despite discontinuation of the antipsychotic drug. Weight Gain: In short-term schizophrenia and bipolar mania trials, there were differences in mean weight gain between SAPHRIS-treated and placebo-treated patients. In a 52 week study, the proportion of patients with an equal to or greater than 7 percent increase in body weight was 14.7 percent. Orthostatic Hypotension and Syncope and Other Hemodynamic Effects: SAPHRIS may induce orthostatic hypotension and syncope. SAPHRIS should be used with caution in patients with known cardiovascular disease, cerebrovascular disease, conditions which would predispose them to hypotension and in the elderly. SAPHRIS should be used cautiously when treating patients who receive treatment with other drugs that can induce hypotension, bradycardia, respiratory or central nervous system depression. Monitoring of orthostatic vital signs should be considered in all such patients, and a dose reduction should be considered if hypotension occurs. Leukopenia, Neutropenia, and Agranulocytosis: In clinical trial and postmarketing experience, events of leukopenia/neutropenia have been reported temporally related to antipsychotic agents, including SAPHRIS. Patients with a pre-existing low white blood cell count (WBC) or a history of leukopenia/neutropenia should have their complete blood count (CBC) monitored frequently during the first few months of therapy and SAPHRIS should be discontinued at the first sign of a decline in WBC in the absence of other causative factors. QT Prolongation: SAPHRIS was associated with increases in QTc interval ranging from 2 to 5 msec compared to placebo. No patients treated with SAPHRIS experienced QTc increases of equal to or greater than 60 msec from baseline measurements, nor did any experience a QTc of equal to or greater than 500 msec. SAPHRIS should be avoided in combination with other drugs known to prolong QTc interval, in patients with congenital prolongation of QT interval or a history of cardiac arrhythmias, and in circumstances that may increase the occurrence of torsades de pointes and/or sudden death in association with the use of drugs that prolong the QTc interval. Hyperprolactinemia: Like other drugs that antagonize dopamine D2 receptors, SAPHRIS can elevate prolactin levels, and the elevation can persist during chronic administration. Galactorrhea, amenorrhea, gynecomastia and impotence have been reported in patients receiving prolactin-elevating compounds. Seizures: SAPHRIS should be used cautiously in patients with a history of seizures or with conditions that lower seizure threshold, e.g., Alzheimer's dementia. Dysphagia: Esophageal dysmotility and aspiration have been associated with antipsychotic drug use. Aspiration pneumonia is a common cause of morbidity and mortality in elderly patients, in particular those with advanced Alzheimer's dementia. SAPHRIS is not indicated for the treatment of dementia-related psychosis, and should not be used in patients at risk for aspiration pneumonia. Potential for Cognitive and Motor Impairment: Somnolence was reported in patients treated with SAPHRIS. Patients should be cautioned about performing activities requiring mental alertness, such as operating hazardous machinery or operating a motor vehicle, until they are reasonably certain that SAPHRIS therapy does not affect them adversely. Suicide: The possibility of suicide attempt is inherent in psychotic illnesses and bipolar disorder. Close supervision of high-risk patients should accompany drug therapy. Prescriptions for SAPHRIS should be written for the smallest quantity of tablets in order to reduce the risk of overdose. SAPHRIS is not recommended in patients with severe hepatic impairment. Co-administration of SAPHRIS with strong CYP1A2 inhibitors (fluvoxamine) or compounds which are both CYP2D6 substrates and inhibitors (paroxetine) should be done with caution. Commonly observed adverse reactions (incidence equal to or greater than 5 percent and at least twice that for placebo) were: Patients with Schizophrenia: akathisia, oral hypoesthesia and somnolence. Patients with Bipolar Disorder: somnolence, dizziness, extrapyramidal symptoms other than akathisia and weight increase. Please see full prescribing information at www.SAPHRIS.com. About Schizophrenia About Bipolar I Disorder About Schering-Plough SCHERING-PLOUGH DISCLOSURE NOTICE: The information in this press release includes certain "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995, including statements relating to the clinical development of, the commercial plans for and the potential market for SAPHRIS/SYCREST. Forward-looking statements relate to expectations or forecasts of future events. Schering-Plough does not assume the obligation to update any forward-looking statement. Many factors could cause actual results to differ materially from Schering-Plough's forward-looking statements, including uncertainties in the regulatory process, among other uncertainties. For further details about these and other factors that may impact the forward-looking statements, see Schering-Plough's Securities and Exchange Commission filings, including Part II, Item 1A. "Risk Factors" in the Company's second quarter 2009 10-Q, filed July 24, 2009. 美国FDA批准先灵葆雅精神病新药SAPHRIS (商品名称:asenapine) 先灵葆雅药物研发部负责人托马斯·凯斯特勒表示:“该药是一种全新的重要疗法,可为那些首次用药治疗或中断了前期用药的患者带来福音。对于先灵葆雅的产品线来说,它是一个重要补充,也是我们得益于与Organon的联合而在美国获准的第一种药物。” 此次SAPHRIS通过FDA批准的依据是一系列临床试验数据,参加试验的受试者人数达3000人,均为精神分裂症患者和双相躁狂症患者。在SAPHRIS新药申请内容中,还包含了一份4500名患者用药的安全性信息,其中一些人的用药时间超过了2年。此外,该药还进行了一项紧急治疗临床实验,期间将SAPHRIS受试组和安慰剂对照组的疗效进行了对比,结果进一步证实了药物的疗效,这也是它获得批准的重要依据之一。SAPHRIS有望于今年第四季度进入美国市场。 |
Asenapine(商品名:Sycrest)用于精神分裂症
责任编辑:admin |
最新图片
推荐图片更多
热点图片更多 |