|
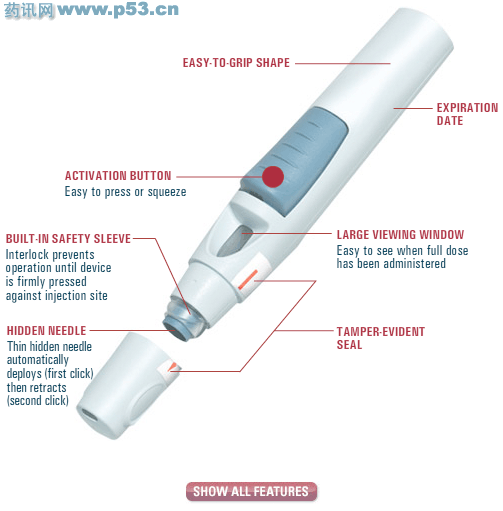
近日,美国食品药品管理局(FDA)批准治疗成人中重度风湿性关节炎、活动型银屑病性关节炎及活动型强直性脊柱炎的长效药物Simponi上市。 Simponi – An Effective New Treatment for Rheumatoid Arthritis, Psoriatic Arthritis, and Ankylosing Spondylitis。 TORONTO — Health Canada issued a Notice of Compliance to Centocor Ortho Biotech Inc on April 7th, 2009 for the biological therapy Simponi – golimumab – which is a tumor necrosis factor α inhibitor for treatment of Rheumatoid Arthritis, Psoriatic Arthritis, and Ankylosing Spondylitis. SIMPONIIndications for SIMPONIModerately to severely active rheumatoid arthritis (RA), in combination with methotrexate (MTX). Active psoriatic arthritis (PsA), alone or with MTX. Active ankylosing spondylitis (AS). Adult dose for SIMPONI50mg SC once monthly. Rotate sites. Children's dosing for SIMPONI<18yrs: not recommended. Warnings/Precautions for SIMPONIIncreased risk of serious or fatal infections (eg, TB, bacterial sepsis, invasive fungal). Active infections: do not initiate therapy. Chronic or history of recurring or opportunistic infections. Conditions that predispose to infection. Travel to, or residence in, areas with endemic TB or mycoses. Test for and treat latent TB prior to initiating therapy. Monitor closely if new infection, reactivation of hepatitis B virus (HBV), or blood dyscrasias develop; discontinue if serious or opportunistic infection, sepsis, HBV reactivation, new or worsening CHF, or hematological abnormality (eg, cytopenias) develops. CHF (monitor). Immunosuppression. CNS demyelinating disorders. Malignancies. Latex allergy. Elderly. Pregnancy (Cat.B). Nursing mothers: not recommended. Interactions for SIMPONIConcurrent abatacept, anakinra, live vaccines, or other TNF blockers: not recommended. Immunosuppressants increase risk of infection. Monitor CYP450 substrates with narrow therapeutic index. Adverse Reactions for SIMPONIInj site reactions, infections (may be serious), upper respiratory tract infection, nasopharyngitis, hypertension; rare: malignancies (eg, lymphoma, esp children), blood dyscrasias, new or worsening CHF, elevated liver enzymes, antibody formation, exacerbation or new onset of psoriasis. How is SIMPONI supplied?Single-dose SmartJect autoinjector—1 |
|
当前位置:药品说明书与价格首页 >> 综合药讯 >> 美国FDA批准治疗关节炎药物Simponi上市
美国FDA批准治疗关节炎药物Simponi上市简介:
近日,美国食品药品管理局(FDA)批准治疗成人中重度风湿性关节炎、活动型银屑病性关节炎及活动型强直性脊柱炎的长效药物Simponi上市。 这3类关节炎均属慢性自身免疫性疾病,可造成患者关节强直、疼痛以 ... 责任编辑:admin |
最新文章更多推荐文章更多热点文章更多
|