|
英文药名: Cytarabine Injection 中文药名: 阿糖胞苷注射剂 药品介绍 【适用症】 【注意事项】 【用法用量】 【禁忌症】孕妇及哺乳期妇女忌用。 【孕妇、哺乳期妇女用药注意事项】孕妇及哺乳期妇女最好不用。 【老年人用药注意事项】由于老年人对化疗药物的耐受性差,用药需减量并注意根据体征等及时调整药量。 【不良反应】 【药物相互作用】 【药理】 【药物代谢动力学】 【用药过量】 规格: 1000mg*100ml CYTARABINE injection, solution
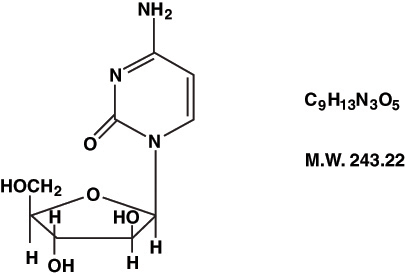
Cytarabine Injection, an antineoplastic, is a sterile isotonic solution for intravenous and subcutaneous use, which contains no preservative and is available in 20 mg/mL (1000 mg/50 mL) Pharmacy Bulk Package. A Pharmacy Bulk Package is a container of a sterile preparation for parenteral use that contains many single doses. The contents are intended for use in a pharmacy admixture program and are restricted to the preparation of admixtures for intravenous infusion. Each mL contains 20 mg Cytarabine, USP and the following inactive ingredients: sodium chloride 0.68% and Water for Injection q.s. When necessary, the pH is adjusted with hydrochloric acid and/or sodium hydroxide to a target pH of 7.4. Each vial contains approximately 5.82 mEq sodium. Cytarabine is chemically 4-amino-1-β-D-arabinofuranosyl-2(1H)-pyrimidinone. The structural formula is:
Cytarabine is an odorless, white to off-white, crystalline powder which is freely soluble in water and slightly soluble in alcohol and in chloroform. CLINICAL PHARMACOLOGY Cell Culture Studies: Cytarabine is cytotoxic to a wide variety of proliferating mammalian cells in culture. It exhibits cell phase specificity, primarily killing cells undergoing DNA synthesis (S-phase) and under certain conditions blocking the progression of cells from the G1 phase to the S-phase. Although the mechanism of action is not completely understood, it appears that cytarabine acts through the inhibition of DNA polymerase. A limited, but significant, incorporation of cytarabine into both DNA and RNA has also been reported. Extensive chromosomal damage, including chromatoid breaks, have been produced by cytarabine and malignant transformation of rodent cells in culture has been reported. Deoxycytidine prevents or delays (but does not reverse) the cytotoxic activity. Cellular Resistance and Sensitivity: Cytarabine is metabolized by deoxycytidine kinase and other nucleotide kinases to the nucleotide triphosphate, an effective inhibitor of DNA polymerase; it is inactivated by a pyrimidine nucleoside deaminase which converts it to the nontoxic uracil derivative. It appears that the balance of kinase and deaminase levels may be an important factor in determining sensitivity or resistance of the cell to cytarabine. Human Pharmacology: Cytarabine is rapidly metabolized and is not effective orally; less than 20 percent of the orally administered dose is absorbed from the gastrointestinal tract. Following rapid intravenous injection of cytarabine, labeled with tritium, the disappearance from plasma is biphasic. There is an initial distributive phase with a half-life of about 10 minutes, followed by a second elimination phase with a half-life of about 1 to 3 hours. After the distributive phase, more than 80 percent of plasma radioactivity can be accounted for by the inactive metabolite 1- β -D-arabinofuranosyluracil (ara-U). Within 24 hours about 80 percent of the administered radioactivity can be recovered in the urine, approximately 90 percent of which is excreted as ara-U. Relatively constant plasma levels can be achieved by continuous intravenous infusion. After subcutaneous or intramuscular administration of cytarabine labeled with tritium, peak-plasma levels of radioactivity are achieved about 20 to 60 minutes after injection and are considerably lower than those after intravenous administration. Cerebrospinal fluid levels of cytarabine are low in comparison to plasma levels after single intravenous injection. However, in one patient in whom cerebrospinal levels were examined after 2 hours of constant intravenous infusion, levels approached 40 percent of the steady state plasma level. With intrathecal administration, levels of cytarabine in the cerebrospinal fluid declined with a first order half-life of about 2 hours. Because cerebrospinal fluid levels of deaminase are low, little conversion to ara-U was observed. Immunosuppressive Action: Cytarabine is capable of obliterating immune responses in man during administration with little or no accompanying toxicity. Suppression of antibody responses to E. coli-VI antigen and tetanus toxoid have been demonstrated. This suppression was obtained during both primary and secondary antibody responses. Cytarabine also suppressed the development of cell-mediated responses such as delayed hypersensitivity skin reaction to dinitrochlorobenzene. However, it had no effect on already established delayed hypersensitivity reactions. Following 5-day courses of intensive therapy with cytarabine, the immune response was suppressed, as indicated by the following parameters: macrophage ingress into skin windows; circulating antibody response following primary antigenic stimulation; lymphocyte blastogenesis with phytohemagglutinin. A few days after termination of therapy there was a rapid return to normal. INDICATIONS AND USAGE Cytarabine in combination with other approved anticancer drugs is indicated for remission induction in acute non-lymphocytic leukemia of adults and children. It has also been found useful in the treatment of acute lymphocytic leukemia and the blast phase of chronic myelocytic leukemia. Intrathecal administration of cytarabine is indicated in the prophylaxis and treatment of meningeal leukemia. CONTRAINDICATIONS Cytarabine Injection is contraindicated in those patients who are hypersensitive to the drug. WARNINGS (See boxed WARNING) Cytarabine is a potent bone marrow suppressant. Therapy should be started cautiously in patients with pre-existing drug-induced bone marrow suppression. Patients receiving this drug must be under close medical supervision and, during induction therapy, should have leucocyte and platelet counts performed daily. Bone marrow examinations should be performed frequently after blasts have disappeared from the peripheral blood. Facilities should be available for management of complications, possibly fatal, of bone marrow suppression (infection resulting from granulocytopenia and other impaired body defenses, and hemorrhage secondary to thrombocytopenia). One case of anaphylaxis that resulted in acute cardiopulmonary arrest and required resuscitation has been reported. This occurred immediately after the intravenous administration of cytarabine. Severe and at times fatal CNS, GI and pulmonary toxicity (different from that seen with conventional therapy regimens of cytarabine) has been reported following some experimental cytarabine dose schedules. These reactions include reversible corneal toxicity, and hemorrhagic conjunctivitis, which may be prevented or diminished by prophylaxis with a local corticosteroid eye drop; cerebral and cerebellar dysfunction, including personality changes, somnolence and coma, usually reversible; severe gastrointestinal ulceration, including pneumatosis cystoides intestinalis leading to peritonitis; sepsis and liver abscess; pulmonary edema, liver damage with increased hyperbilirubinemia; bowel necrosis; and necrotizing colitis. Rarely, severe skin rash, leading to desquamation has been reported. Complete alopecia is more commonly seen with experimental high dose therapy than with standard cytarabine treatment programs. If experimental high dose therapy is used, do not use a diluent containing benzyl alcohol. Cases of cardiomyopathy with subsequent death has been reported following experimental high dose therapy with cytarabine in combination with cyclophosphamide when used for bone marrow transplant preparation. A syndrome of sudden respiratory distress, rapidly progressing to pulmonary edema and radiographically pronounced cardiomegaly has been reported following experimental high dose therapy with cytarabine used for the treatment of relapsed leukemia from one institution in 16/72 patients. The outcome of this syndrome can be fatal. Two patients with childhood acute myelogenous leukemia who received intrathecal and intravenous cytarabine at conventional doses (in addition to a number of other concomitantly administered drugs) developed delayed progressive ascending paralysis resulting in death in one of the two patients. Use in Pregnancy (Category D): Cytarabine Injection can cause fetal harm when administered to a pregnant woman. Cytarabine causes abnormal cerebellar development in the neonatal hamster and is teratogenic to the rat fetus. There are no adequate and well-controlled studies in pregnant women. Women of childbearing potential should be advised to avoid becoming pregnant. PRECAUTIONS 1. General Precautions: Patients receiving Cytarabine Injection must be monitored closely. Frequent platelet and leukocyte counts and bone marrow examinations are mandatory. Consider suspending or modifying therapy when drug-induced marrow depression has resulted in a platelet count under 50,000 or a polymorphonuclear granulocyte count under 1000/mm3. Counts of formed elements in the peripheral blood may continue to fall after the drug is stopped and reach lowest values after drug-free intervals of 12 to 24 days. When indicated, restart therapy when definite signs of marrow recovery appear (on successive bone marrow studies). Patients whose drug is withheld until “normal” peripheral blood values are attained may escape from control. When large intravenous doses are given quickly, patients are frequently nauseated and may vomit for several hours postinjection. This problem tends to be less severe when the drug is infused. The human liver apparently detoxifies a substantial fraction of an administered dose. In particular, patients with renal or hepatic function impairment may have a higher likelihood of CNS toxicity after high-dose cytarabine treatment. Use the drug with caution and possibly at reduced dose in patients whose liver or kidney function is poor. Periodic checks of bone marrow, liver and kidney functions should be performed in patients receiving Cytarabine Injection. Like other cytotoxic drugs, Cytarabine Injection may induce hyperuricemia secondary to rapid lysis of neoplastic cells. The clinician should monitor the patient’s blood uric acid level and be prepared to use such supportive and pharmacologic measures as might be necessary to control this problem. Acute pancreatitis has been reported to occur in a patient receiving cytarabine by continuous infusion and in patients being treated with cytarabine who have had prior treatment with L-asparaginase. 2. Information for patient: Not applicable. 3. Laboratory tests: See General Precautions. 4. Drug Interactions: Reversible decreases in steady-state plasma digoxin concentrations and renal glycoside excretion were observed in patients receiving beta-acetyldigoxin and chemotherapy regimens containing cyclophosphamide, vincristine and prednisone with or without cytarabine or procarbazine. Steady-state plasma digitoxin concentrations did not appear to change. Therefore, monitoring of plasma digoxin levels may be indicated in patients receiving similar combination chemotherapy regimens. The utilization of digitoxin for such patients may be considered as an alternative. An in vitro interaction study between gentamicin and cytarabine showed a cytarabine related antagonism for the susceptibility of K. pneumoniae strains. This study suggests that in patients on cytarabine being treated with gentamicin for a K. pneumoniae infection, the lack of a prompt therapeutic response may indicate the need for reevaluation of antibacterial therapy. Clinical evidence in one patient showed possible inhibition of fluorocytosine efficacy during therapy with cytarabine. This may be due to potential competitive inhibition of its uptake. 5. Carcinogenesis, mutagenesis, impairment of fertility: Extensive chromosomal damage, including chromatoid breaks have been produced by cytarabine and malignant transformation of rodent cells in culture has been reported. 6. Pregnancy: Pregnancy Category D. See WARNINGS. A review of the literature has shown 32 reported cases where cytarabine was given during pregnancy, either alone or in combination with other cytotoxic agents. Eighteen normal infants were delivered. Four of these had first trimester exposure. Five infants were premature or of low birth weight. Twelve of the 18 normal infants were followed up at ages ranging from six weeks to seven years, and showed no abnormalities. One apparently normal infant died at 90 days of gastroenteritis. Two cases of congenital abnormalities have been reported, one with upper and lower distal limb defects, and the other with extremity and ear deformities. Both of these cases had first trimester exposure. There were seven infants with various problems in the neonatal period, including pancytopenia; transient depression of the WBC, hematocrit or platelets; electrolyte abnormalities; transient eosinophilia; and one case of increased IgM levels and hyperpyrexia possibly due to sepsis. Six of the seven infants were also premature. The child with pancytopenia died at 21 days of sepsis. Therapeutic abortions were done in five cases. Four fetuses were grossly normal, but one had an enlarged spleen and another showed Trisomy C chromosome abnormality in the chorionic tissue. Because of the potential for abnormalities with cytotoxic therapy, particularly during the first trimester, a patient who is or who may become pregnant while on Cytarabine Injection should be apprised of the potential risk to the fetus and the advisability of pregnancy continuation. There is a definite, but considerably reduced risk if therapy is initiated during the second or third trimester. Although normal infants have been delivered to patients treated in all three trimesters of pregnancy, follow-up of such infants would be advisable. 7. Labor and delivery: Not applicable. 8. Nursing mothers: It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from cytarabine, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother. 9. Pediatric use: See INDICATIONS AND USAGE. ADVERSE REACTIONS Expected Reactions: Because cytarabine is a bone marrow suppressant, anemia, leukopenia, thrombocytopenia, megaloblastosis and reduced reticulocytes can be expected as a result of administration with cytarabine. The severity of these reactions are dose and schedule dependent. Cellular changes in the morphology of bone marrow and peripheral smears can be expected. Following 5-day constant infusions or acute injections of 50 mg/m2 to 600 mg/m2, white cell depression follows a biphasic course. Regardless of initial count, dosage level, or schedule, there is an initial fall starting the first 24 hours with a nadir at days 7-9. This is followed by a brief rise which peaks around the twelfth day. A second and deeper fall reaches nadir at days 15-24. Then there is a rapid rise to above baseline in the next 10 days. Platelet depression is noticeable at 5 days with a peak depression occurring between days 12-15. Thereupon, a rapid rise to above baseline occurs in the next 10 days. Infectious Complications: Infection: Viral, bacterial, fungal, parasitic, or saprophytic infections, in any location in the body may be associated with the use of cytarabine alone or in combination with other immunosuppressive agents following immunosuppressant doses that affect cellular or humoral immunity. These infections may be mild, but can be severe and at times fatal. The Cytarabine (Ara-C) Syndrome: A cytarabine syndrome has been described by Castleberry. It is characterized by fever, myalgia, bone pain, occasionally chest pain, maculopapular rash, conjunctivitis and malaise. It usually occurs 6-12 hours following drug administration. Corticosteroids have been shown to be beneficial in treating or preventing this syndrome. If the symptoms of the syndrome are deemed treatable, corticosteroids should be contemplated as well as continuation of therapy with Cytarabine Injection. Most Frequent Adverse Reactions: Anorexia, hepatic dysfunction, nausea, fever, vomiting, rash, diarrhea, thrombophlebitis, oral and anal inflammation or ulceration, bleeding (all sites). Nausea and vomiting are most frequent following rapid intravenous injection. Less Frequent Adverse Reactions: Sepsis, abdominal pain, pneumonia, freckling, cellulitis at injection site, jaundice, skin ulceration, conjunctivitis (may occur with rash), urinary retention, dizziness, renal dysfunction, alopecia, neuritis, anaphylaxis (see WARNINGS), neural toxicity, allergic edema, sore throat, pruritus, esophageal ulceration, shortness of breath, esophagitis, urticaria, chest pain, pericarditis, headache, bowel necrosis, pancreatitis. Experimental Doses: Severe and at times fatal CNS, GI and pulmonary toxicity (different from that seen with conventional therapy regimens of cytarabine) has been reported following some experimental dose schedules of Cytarabine. These reactions include reversible corneal toxicity and hemorrhagic conjunctivitis, which may be prevented or diminished by prophylaxis with a local corticosteroid eye drop; cerebral and cerebellar dysfunction, including personality changes, somnolence and coma, usually reversible; severe gastrointestinal ulceration, including pneumatosis cystoides intestinalis leading to peritonitis; sepsis and liver abscess; pulmonary edema, liver damage with increased hyperbilirubinemia; bowel necrosis; and necrotizing colitis. Rarely, severe skin rash, leading to desquamation has been reported. Complete alopecia is more commonly seen with experimental high dose therapy than with standard cytarabine treatment programs. If experimental high dose therapy is used, do not use a diluent containing benzyl alcohol. Cases of cardiomyopathy with subsequent death has been reported following experimental high dose therapy with cytarabine in combination with cyclophosphamide when used for bone marrow transplant preparation. This cardiac toxicity may be schedule dependent. A syndrome of sudden respiratory distress, rapidly progressing to pulmonary edema and radiographically pronounced cardiomegaly has been reported following experimental high dose therapy with cytarabine used for the treatment of relapsed leukemia from one institution in 16/72 patients. The outcome of this syndrome can be fatal. Two patients with adult acute non-lymphocytic leukemia developed peripheral motor and sensory neuropathies after consolidation with high-dose cytarabine, daunorubicin, and asparaginase. Patients treated with high-dose cytarabine should be observed for neuropathy since dose schedule alterations may be needed to avoid irreversible neurologic disorders. Ten patients treated with experimental intermediate doses of cytarabine (1 g/m2) with and without other chemotherapeutic agents (meta-AMSA, daunorubicin, etoposide) at various dose regimens developed a diffuse interstitial pneumonitis without clear cause that may have been related to the cytarabine. Two cases of pancreatitis have been reported following experimental doses of cytarabine and numerous other drugs. Cytarabine could have been the causative agent. OVERDOSAGE There is no antidote for cytarabine overdosage. Doses of 4.5 g/m2 by intravenous infusion over 1 hour every 12 hours for 12 doses has caused an unacceptable increase in irreversible CNS toxicity and death. Single doses as high as 3 g/m2 have been administered by rapid intravenous infusion without apparent toxicity. DOSAGE AND ADMINISTRATION Cytarabine Injection (non-preserved) can be administered by intravenous injection or infusion, subcutaneously, or intrathecally. However, the intent of this Pharmacy Bulk Package is for the preparation of solutions for IV infusion only. Intrathecal use of cytarabine requires the use of single-dose, unpreserved solutions only. Cytarabine Injection is not active orally. The schedule and method of administration varies with the program of therapy to be used. While Cytarabine Injection may be given by intravenous infusion or injection, or subcutaneously or intrathecally, THE PURPOSE OF THE PHARMACY BULK PACKAGE IS FOR THE PREPARATION OF INTRAVENOUS INFUSIONS. Thrombophlebitis has occurred at the site of drug injection or infusion in some patients, and rarely patients have noted pain and inflammation at subcutaneous injection sites. In most instances, however, the drug has been well tolerated. Patients can tolerate higher total doses when they receive the drug by rapid intravenous injection as compared with slow infusion. This phenomenon is related to the drug’s rapid inactivation and brief exposure of susceptible normal and neoplastic cells to significant levels after rapid injection. Normal and neoplastic cells seem to respond in somewhat parallel fashion to these different modes of administration and no clear-cut clinical advantage has been demonstrated for either. In the induction therapy of acute non-lymphocytic leukemia, the usual cytarabine dose in combination with other anti-cancer drugs is 100 mg/m2/day by continuous IV infusion (Days 1-7) or 100 mg/m2 IV every 12 hours (Days 1-7). The literature should be consulted for the current recommendations for use in acute lymphocytic leukemia. Intrathecal Use in Meningeal Leukemia: Cytarabine has been used intrathecally in acute leukemia in doses ranging from 5 mg/m2 to 75 mg/m2 of body surface area. The frequency of administration varied from once a day for 4 days to once every 4 days. The most frequently used dose was 30 mg/m2 every 4 days until cerebrospinal fluid findings were normal, followed by one additional treatment. The dosage schedule is usually governed by the type and severity of central nervous system manifestations and the response to previous therapy. If used intrathecally, do not use a solution containing benzyl alcohol. This pharmacy bulk package is not intended to be used for the preparation of intrathecal doses. Cytarabine given intrathecally may cause systemic toxicity and careful monitoring of the hemopoietic system is indicated. Modification of other anti-leukemia therapy may be necessary. Major toxicity is rare. The most frequently reported reactions after intrathecal administration were nausea, vomiting and fever; these reactions are mild and self-limiting. Paraplegia has been reported. Necrotizing leukoencephalopathy occurred in 5 children; these patients had also been treated with intrathecal methotrexate and hydrocortisone, as well as by central nervous system radiation. Isolated neurotoxicity has been reported. Blindness occurred in two patients in remission whose treatment had consisted of combination systemic chemotherapy, prophylactic central nervous system radiation and intrathecal cytarabine. When cytarabine is administered both intrathecally and intravenously within a few days, there is an increased risk of spinal cord toxicity, however, in serious life-threatening disease, concurrent use of intravenous and intrathecal cytarabine is left to the discretion of the treating physician. Focal leukemic involvement of the central nervous system may not respond to intrathecal cytarabine and may better be treated with radiotherapy. Chemical Stability of Infusion Solutions: Chemical stability studies were performed by a stability indicating HPLC assay on Cytarabine Injection in infusion solutions. These studies showed that when Cytarabine Injection was diluted with Water for Injection, 5% Dextrose Injection or Sodium Chloride Injection, in both glass and plastic infusion bags, 97-100% of the cytarabine was present after 8 days storage at room temperature. This chemical stability information in no way indicates that it would be acceptable practice to infuse a cytarabine admixture well after the preparation time. Good professional practice suggests that administration of an admixture should be as soon after preparation as feasible. Parenteral drugs should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Handling and Disposal: Procedures for proper handling and disposal of anti-cancer drugs should be considered. Several guidelines on this subject have been published.1-7 There is no general agreement that all of the procedures recommended in the guidelines are necessary or appropriate. Direction for Dispensing From Pharmacy Bulk Package: The 50 mL Pharmacy Bulk Package is for use in the Pharmacy Admixtures Service only. The vials should be inserted into the plastic handling device provided, suspended as a unit in the laminar flow hood. A single entry through the vial closure should be made with a sterile dispensing set or transfer device. Transfer individual doses to appropriate intravenous infusion solutions. Use of a syringe with needle is not recommended. Multiple entries will increase the potential of microbial and particulate contamination. The above process should be carried out under a laminar flow hood using aseptic technique. Care should be exercised to protect personnel from aerosolized drug (see DOSAGE AND ADMINISTRATION, REFERENCES). Discard any unused portion within 4 hours after initial closure entry. HOW SUPPLIED Cytarabine Injection, PHARMACY BULK PACKAGE. Sterile, Isotonic Solution. Preservative Free. NDC No. 61703-303-46. Cytarabine Injection 20 mg/mL (1000 mg) in a 50 mL flip-top vial (green cap), packaged individually. Store product at controlled room temperature, 15° - 30°C (59° - 86°F). |
阿糖胞苷注射剂(Cytarabine Injection)简介:
英文药名: Cytarabine Injection
中文药名: 阿糖胞苷注射剂
药品介绍
【适用症】 注射用盐酸阿糖胞苷适用于急性白血病的诱导缓解期及维持巩固期。对急性非淋巴细胞性白血病效果较好,对慢性粒细胞 ... 责任编辑:admin |
最新文章更多推荐文章更多热点文章更多 |